Polar bond
In chemistry, a polar bond is a type of covalent bond between two or more dissimilar atoms, in which electrons are shared unequally. A covalent bond is the "strong" kind of bond that makes a molecule.[1]
Electrons are negatively charged. If the two atoms share the electrons equally, then the negative charge is evenly spread out over the entire molecule, balanced out by the positive charges of the protons in the atoms' nuclei. This makes the entire molecule neutral - a non-polar bond, making a non-polar molecule. However, if one of the atoms is particularly greedy for electrons (called electronegativity), then its side of the bond will have more electrons. Therefore, that side of the bond will have a slightly negative charge, and the other side will have a slightly positive charge.[2]
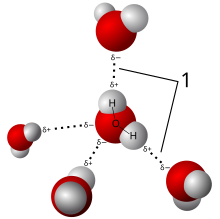
This is important because it turns the bond into an electrical dipole. This dipole acts like a magnet, if other bonds that work as dipoles are nearby, it will react to them.
In some molecules, these polar bonds are lined up with each other. Then the entire molecule is a dipole, and attracts other dipoles. Water does this. You can see in the image that the hydrogen (small and white) is positively charged and the oxygen (big and red) is negatively charged. This picture shows how the little water dipoles line up with each other, in the same way that if you toss a bunch of little magnets into a box, they will line up with each other.[3]
This is one of the most basic and important differences between bonds (and the resulting molecules) in chemistry. This can be easily predicted (because the electronegativity of all the atoms in the periodic table is known). It creates an enormous difference in how the molecules behave.
When the difference in electron negativity between the 2 atoms Is more in covalent bond the more electron negativity, atom acquire partial negativity charge and other atom acqiure partial positive charge.
Related pages[change | change source]
References[change | change source]
