Aldol reaction
The aldol reaction is a very important reaction in organic chemistry. It allows to form new carbon-carbon bonds. It was discovered in 1872.
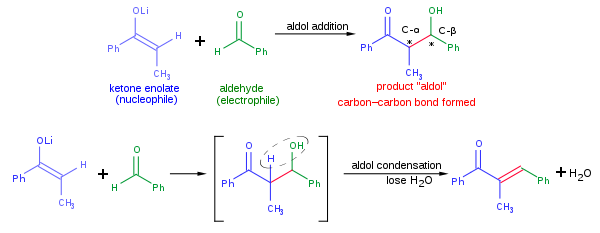
The reagents are two carbonyl compounds together with a base. A proton is removed from one of them forming a negative charge on the α-carbon (the carbon just next to the C-O double bond). The negative charge can then attack the other carbonyl compound, forming the bond. The product is a β-hydroxy carbonyl compound, a molecule with a C-O double bond and an alcohol two carbon atoms down the chain. This reaction is very powerful because it can form a big molecule from two smaller ones.
Modern ways of doing the aldol reaction also allow to control the stereochemistry of the product. This is a very good way to create new chiral centres.
References[change | change source]
- Clayden, Jonathan; Greeves, Nick; Warren, Stuart (2012). Organic Chemistry (2nd ed.). Oxford University Press. pp. 614-655. ISBN 9780199270293.
