Capsaicin
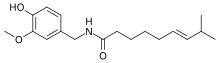

Capsaicin is a chemical substance. It is responsible for the sense of spiciness or hotness found in chili peppers. In mammals, it causes a sensation of burning of the tissues it comes in contact with.[1] Capsaicin, and other similar substances called capsaicinoids are produced by chili peppers and other plants, probably as a protection against being eaten.[2] Pure capsaicin is a hydrophobic, colorless, odorless, crystalline to waxy compound.
Capsaicinoids are added to food to make it have a hot taste, but it can also be used as an analgesic, a painkiller. Such painkillers are often directly used on the skin. The burning of the capsaicin will mask the real pain. Capsaicin is also the main agent in pepper spray.
Capsaicin is not soluble in water; it binds to oil and fat. Soap can be used to wash it off. If something too hot has been eaten, milk or yoghurt can be used.
References[change | change source]
- ↑ It is likely that the effects work on any other kind of herbivore, but on mammals it certainly does.
- ↑ What made chili peppers so spicy? Talk of the Nation, 15 Aug 2008.
