Conrotatory and disrotatory
These terms describe two classes of an electrocyclic reaction (a type of organic chemical reactions). In a conrotatory mode, the substituents located at the ends of a conjugated double bond system move in the same (clockwise or counterclockwise) direction during ring opening or ring closure. In a disrotatory mode, they move in opposite directions.
An example is the conversion of trans-cis-trans-2,4,6-octatriene to cis-dimethylcyclohexadiene (top of figure). The orbital mechanics of the reaction needs a disrotatory mode. The orbital symmetry of the octatriene's highest occupied molecular orbital (HOMO) requires for the end pi orbitals to move in opposite directions to form the correct symmetry found in the sigma bond.
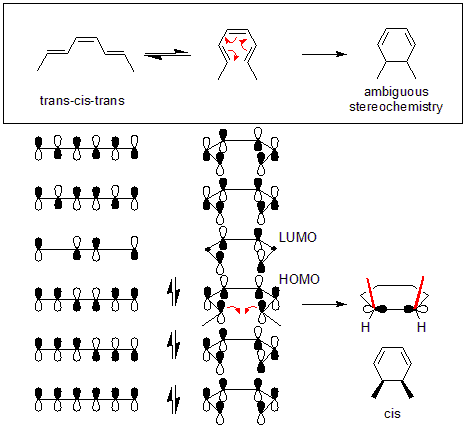
Thermal rearrangements of all conjugated systems containing 4n + 2 pi electron are stereospecific. This is based on preservation of orbital symmetry in the highest occupied molecular orbital. Systems containing 4n pi electrons show the opposite conrotatory mode. This is also true for rearrangements of 4n + 2 pi (where n is a whole number) electrons driven by light (photoinduced). Photoinduced rearrangements of 4n pi electron systems (where the number of electrons is divisible by 4) follow the disrotatory rule.
The Woodward–Hoffmann rules summarize the above different reactions.
The following picture also shows the difference between conrotatory and disrotatory reactions:

References[change | change source]
- Carey, Francis A.; Sundberg, Richard J.; (1984). Advanced Organic Chemistry Part A Structure and Mechanisms (2nd ed.). New York N.Y.: Plenum Press. ISBN 0-306-41198-9.
- March Jerry; (1985). Advanced Organic Chemistry reactions, mechanisms and structure (3rd ed.). New York: John Wiley & Sons, inc. ISBN 0-471-85472-7
