Polyester
Polyester is a type of man-made material. It is a synthetic polymer. With an 18% market share of all plastic materials produced, it is third after polyethylene (33.5%) and polypropylene (19.5%).
Uses
[change | change source]Polyester can be made into thread, yarn or clothes. Woven into synthetic fabrics,[1] they are often used to make clothes and home furnishings. These include shirts, trousers, pants, jackets, hats, bedding and upholstered furniture. Polyester fiber is used as cushioning and insulating material in pillows, comforters and upholstery padding. It can be used in curtains to insulate windows and keep heat indoors. Polyester is used to make insulating carpets.
Industrial polyester fibers, yarns and ropes are used in tires, fabrics for their use, and safety belts.
Chemistry
[change | change source]
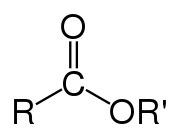
Polyester is a type of polymer that has the ester functional group in its main chain. There are many different polyesters. The term "polyester" as a specific material most often refers to polyethylene terephthalate (PET). Polyesters include chemicals found in nature, such as in plant cuticles. Synthetics made by step-growth polymerization such as polycarbonate and polybutyrate are also polyesters. Natural polyesters and a few synthetic ones are biodegradable, but most synthetic polyesters are not.
The most common polyesters are thermoplastics.[2]
Synthesis
[change | change source]Most polyesters are made with a polycondensation reaction. See "condensation reactions in polymer chemistry". The general equation for the reaction of a diol with a diacid is :
- (n+1) R(OH)2 + n R´(COOH)2 → HO[ROOCR´COO]nROH + 2n pH2O
An alcohol and a carboxylic acid react to form a carboxylic ester. To assemble a polymer, the water formed by the reaction must be continually removed by azeotrope distillation.
References
[change | change source]- ↑ Vải polyester là gì [What is polyester fabric] (in Vietnamese). DANANGSALE. 3 July 2020.
- ↑ Rosato, Dominick V.; Rosato, Donald V.; Rosato, Matthew V. (2004), Plastic product material and process selection handbook, Elsevier, p. 85, ISBN 9781856174312
