Neutron
Neutrons, with protons and electrons, make up an atom. Neutrons and protons are found in the nucleus of an atom.[1][2][3] Unlike protons, which have a positive charge, or electrons, which have a negative charge, neutrons have zero charge[1][4] which means they are neutral particles. Neutrons bind with protons with the residual strong force.
Neutrons were predicted by Ernest Rutherford,[5] and discovered by James Chadwick,[6][7] in 1932.[6] Atoms were fired at a thin pane of beryllium. Particles emerged which had no charge, and he called these 'neutrons'. They were later added to the modern image of the atom.
Neutrons have a mass of 1.675 × 10-24g,[8] which is a little heavier than the proton.[8] Neutrons are 1839 times heavier than electrons.[8]
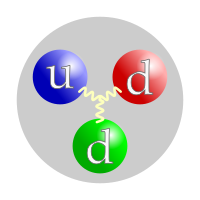
Like all hadrons, neutrons are made of quarks. A neutron is made of two down quarks and one up quark.[2][3] One up quark has a charge of +2/3, and the two down quarks each have a charge of -1/3. The fact that these charges cancel out is why neutrons have a neutral (0) charge. Quarks are held together by gluons.
Isotopes[change | change source]
Neutrons can be found in almost all atoms together with protons and electrons. Hydrogen-1 is the only exception. Atoms with the same number of protons but a different number of neutrons are called isotopes of the same element.
The number of neutrons in an atom does not affect its chemical properties. However it affects its half-life, a measure of its stability. An unstable isotope has a short half-life, in which half of it decays to lighter elements. By contrast, a stable isotope has a long half-life, much longer than that of an unstable isotope. The stability of an isotope is related to radioactivity: an unstable isotope can be highly radioactive.
Atomic reactions[change | change source]
Neutrons are the key to nuclear chain reactions, nuclear power and nuclear weapons.
Related pages[change | change source]
References[change | change source]
- ↑ 1.0 1.1 Woolley, Steve (2011). Edexcel IGCSE Physics Revision Guide. Pearson Education. p. 20-21. ISBN 9780435046736.
- ↑ 2.0 2.1 Cox, Brian; Cohen, Andrew (2011). Wonders of the Universe. HarperCollins. p. 79, 108. ISBN 9780007395828.
- ↑ Ryan, Lawrie (2001). Chemistry for you: revised National Curriculum edition for GCSE (Second ed.). Nelson Thornes. p. 29. ISBN 9780748762347.
- ↑ "Ernest Rutherford". chemed.chem.purdue.edu.
- ↑ 6.0 6.1 "Discovery of Neutrons". Helmholtz Zentrum Berlin. 2008-08-23. Archived from the original on 2010-11-13. Retrieved 2011-04-14.
- ↑ "The Nobel Prize in Physics 1935: James Chadwick". Nobelprize.org. Retrieved 2011-04-15.
- ↑ 8.0 8.1 8.2 "Neutron (subatomic particle)". Encyclopædia Britannica Online. Encyclopædia Britannica. Retrieved 2011-04-14.
