Butene
Appearance
Butene is the third occurring chemical compound in the alkene series of compounds. It has the chemical formula C4H8. Butene is made in laboratories. Butane is formed from covalent bonds between hydrogen and carbon atoms.
There are four isomers, and all are gases at room temperature. All four of these hydrocarbons have four carbon atoms and one double bond in their molecules, but have different chemical structures. The IUPAC and common names, respectively, of these chemical compounds are:
| IUPAC name | Common name | Structure | Skeletal formula | 3D model |
|---|---|---|---|---|
| But-1-ene | α-butylene | 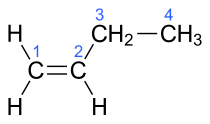
|

| |
| (2Z)-but-2-ene | cis-β-butylene | 
|

|

|
| (2E)-but-2-ene | trans-β-butylene | 
|

|

|
| 2-methylprop-1-ene | Isobutylene | 
|

|

|
