Condensation

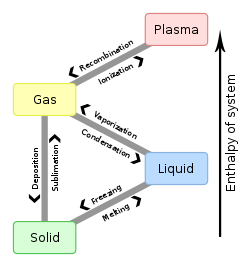
Condensation is how gas changes into a liquid when it touches a cooler surface. Condensation is part of the water cycle. It is the opposite of evaporation.[1]
Process[change | change source]
Condensation of water is when water changes from gas to a liquid or crystal shape. Any gas can condense, either because the temperature is dropping or the pressure is increasing. In either case the pressure of the condensing gas is higher than the vapor pressure of the gas, at that temperature).[2]
During condensation, the molecules in the matter slow down. Heat energy is taken away, causing the state of matter to change. Condensation is exothermic. This means that condensation makes the temperature go up. Evaporation is the opposite and causes a temperature loss.
It is a useful process. One use is in distillation. The gas made from a boiling liquid mix is sent to a condenser, where the different parts condense differently. This can purify a liquid, such as alcohol or water. The condensing liquid makes heat, which must be removed for the condensation to continue.
Related pages[change | change source]
References[change | change source]
- ↑ International Union of Pure and Applied Chemistry. "condensation in atmospheric chemistry". Compendium of Chemical Terminology Internet edition.
- ↑ "condensation in atmospheric chemistry". The IUPAC Compendium of Chemical Terminology. 2014. doi:10.1351/goldbook.C01235.
Other websites[change | change source]
- International Consortium of Advanced Technologies and Security at SDSU Archived 2007-09-11 at the Wayback Machine
- ScienceDirect - Heat transfer and evaporation in geothermal desalination units
- Condensation (phase transition) -Citizendium