Enthalpy of fusion
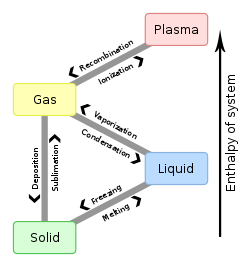
Enthalpy of fusion is the measure of the energy needed to change a substance from a solid to a liquid. Extra energy is needed, more than what is obvious by the temperature scale. For example, ice at 0°C needs to absorb heat before it converts (changes) to water at 0°C. The temperature does not change, but energy is absorbed to change from solid to liquid.
Related pages[change | change source]