Protein folding

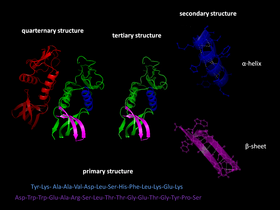
Protein folding is how a protein gets its functional shape or 'conformation'. It is mainly a self-organising process.[1] Starting from a random coil, polypeptides fold into their characteristic working shape.[2] The structure is held together by hydrogen bonds.
The stages are:
- Each protein exists as an unfolded polypeptide or random coil when translated from a sequence of mRNA to a linear chain of amino acids. This polypeptide lacks any developed three-dimensional structure (left hand side of the top figure).
- Amino acids interact with each other to produce a well-defined three-dimensional structure, the folded protein (right hand side of the figure). This is known as the native state. The resulting three-dimensional structure is determined by the amino acid sequence (Anfinsen's dogma).[3]
Without its correct three-dimensional structure a protein does not work. However, some parts of proteins may not fold: this is normal.[4]
If proteins do not fold into their native shape, they are inactive and are usually toxic. Several diseases may be caused by misfolded proteins.[5] Many allergies are caused by the folding of the proteins, for the immune system does not produce antibodies for all possible protein structures.[6]
On 30 November 2020, the protein folding was solved by artificial intelligence company DeepMind.[7][8]
Chaperones[change | change source]
Chaperonins are large proteins which help the folding of some proteins after synthesis.[9] Chaperones in general were first discovered helping histones and DNA join up to form nucleosomes.[10] Nucleosomes are the building blocks for chromosomes. This is the way many cell organelles are built up.[11][12]
References[change | change source]
- ↑ Dobson C.M. 2000. The nature and significance of protein folding. In Pain R.H. (ed) Mechanisms of protein folding. Oxford University Press, 1–28. ISBN 0-19-963789-X
- ↑ Alberts, Bruce; et al. (2002). "The shape and structure of proteins". Molecular biology of the cell. New York: 4th ed, Garland Science. ISBN 0-8153-3218-1.
- ↑ Anfinsen C. (1972). "The formation and stabilization of protein structure". Biochem. J. 128 (4): 737–49. doi:10.1042/bj1280737. PMC 1173893. PMID 4565129.
- ↑ Berg, Jeremy M; Tymoczko, John L. & Stryer, Lubert. Web content by Neil D. Clarke (2002). "3. Protein structure and function". Biochemistry. San Francisco: W.H. Freeman. ISBN 0-7167-4684-0.
{{cite book}}: CS1 maint: multiple names: authors list (link) - ↑ Selkoe, Dennis J. (2003). "Folding proteins in fatal ways". Nature. 426 (6968): 900–904. Bibcode:2003Natur.426..900S. doi:10.1038/nature02264. PMID 14685251. S2CID 6451881.
- ↑ Alberts, Bruce et al 2010. Protein structure and function. In Essential cell biology. 3rd ed, New York: Garland Science, 120-170.
- ↑ "DeepMind AI cracks 50-year-old problem of protein folding". The Guardian. 30 November 2020. Retrieved 30 November 2020.
- ↑ "AlphaFold: a solution to a 50-year-old grand challenge in biology". DeepMind. 30 November 2020. Retrieved 30 November 2020.
- ↑ Hartl F.U. 1996. Molecular chaperones in cellular protein folding. Nature 381, 571–579
- ↑ Ellis R.J. 1996. Discovery of molecular chaperones. Cell stress chaperones 1 (3): 155–60.
- ↑ Bartlett A.L. & Radford S.E. 2009. An expanding arsenal of experimental methods yields an explosion of insights into protein folding mechanisms. Nat. Struct. Mol. Biol. 16, 582–588
- ↑ Hartl F.U. & Hayer-Hartl M. 2009. Converging concepts of protein folding in vitro and in vivo. Nature Structural & Molecular Biology 16 (6): 574–581. [1]
