Hepatitis C
This article needs to be updated. (January 2024) |
| Hepatitis C | |
|---|---|
| Classification and external resources | |
 | |
| ICD-10 | B17.1, B18.2 |
| ICD-9 | 070.70,070.4, 070.5 |
| OMIM | 609532 |
| DiseasesDB | 5783 |
| MedlinePlus | 000284 |
| eMedicine | med/993 ped/979 |
| MeSH | D006526 |
Hepatitis C is an infection that mostly affects the liver. The hepatitis C virus (HCV) causes this disease.[1] Often, a person with Hepatitis C does not have any health problems or signs that they have the disease. However, chronic infection can scar the liver. Many years of infection may cause cirrhosis. Sometimes, people with cirrhosis also have liver failure or liver cancer. They can also have very swollen veins of the esophagus and stomach. The blood loss from this problem can kill.[1]
Hepatitis C is usually spread by blood-to-blood contact, when blood from a person with Hepatitis C touches or gets into another person's bloodstream. The most common ways that this happens are through intravenous drug use (when a person shoots drugs into one of their veins, with a needle that was already used by a person infected with Hepatitis C); nonsterile medical equipment (medical tools that were not cleaned well enough after being used on an infected person); and blood transfusions (when a person is given blood that came from an infected person).
Around the world, about 130–170 million people have Hepatitis C. Scientists began studying the Hepatitis C virus in the 1970s, and in 1989 they proved that the virus exists.[2] As far as scientists know, this virus does not cause disease in any animals other than humans.
The medications that are normally used to treat Hepatitis C are called peginterferon and ribavirin. Between 50 and 80% of people who are treated are cured. However, if a person's Hepatitis C has got worse so much that the person has cirrhosis or liver cancer, the person might need a liver transplant (they might need to have surgery where they are given another person's liver, or part of another person's liver). This makes it possible for the person to survive, but the Hepatitis C virus usually comes back after the transplant.[3] There is no vaccine that works to prevent people from getting Hepatitis C.
Signs and symptoms[change | change source]
Hepatitis C causes symptoms that begin quickly or last only a short time in just 15% of people with the disease.[4] More often, infected people have symptoms that are not serious and not very specific, like not feeling like eating, feeling tired, nausea (feeling like throwing up), pain in the muscles or joints, and losing weight.[5] Every once in a while, an infected person may get jaundice where a person's skin turns yellow, a sign that their liver is not working quite right.[6] If it is not treated, Hepatitis C goes away by itself in 10-50% of infected people (1 to 5 out of every 10). This happens more often in young women than in other infected people.[6]
Chronic infection[change | change source]
Eighty percent (or 8 out of every 10) of people exposed to the Hepatitis C virus get a chronic infection (one that does not get better and lasts for a long time).[7] Most experience very few or no symptoms during the first decades of the infection,[8] although chronic Hepatitis C can cause feeling tired.[9] Hepatitis C leads to cirrhosis in 10–30% of people who have been infected over 30 years.[3][5] People with Hepatitis C are more likely to get cirrhosis if they are men; if they are alcoholics; or if they also have Hepatitis B or HIV.[5] Cirrhosis can cause serious problems on its own, but it also makes people more likely to get other serious illnesses. For example, people who get cirrhosis are twenty times more likely to get liver cancer (with about 1-3% getting liver cancer every year).[3][5] People with Hepatitis C who are alcoholics are even more likely - 100 times more likely - to get liver cancer.[10] Among people in general, 27% of all cases of cirrhosis, and 25% of all cases of liver cancer, are caused by Hepatitis C.[11]
Cirrhosis of the liver can cause many different symptoms. Some of these symptoms are high blood pressure in the veins that travel to the liver; a buildup of fluid in the abdomen, called ascites; easy bruising or bleeding; veins becoming larger than normal, especially in the stomach and esophagus; jaundice (a yellowing of the skin); and brain damage.[12]
Effects outside the liver[change | change source]
Hepatitis C can also cause some problems which do not happen very often which affect parts of the body outside of the liver. One rare problem that Hepatitis C can cause is Sjögren's syndrome, an autoimmune disorder where the body's system of defenses attacks itself. Hepatitis C can also cause a lower-than-normal number of blood platelets (the part of the blood which causes blood to clot) without enough platelets, a person can have bleeding problems, or can start bleeding and be unable to stop. Other rare problems that Hepatitis C can cause are long-lasting skin disease; non-Hodgkin's lymphoma (a type of cancer); and diabetes (where a person's body does not make or use enough insulin, an important hormone that controls the level of sugar in the blood).[13][14]
Cause[change | change source]
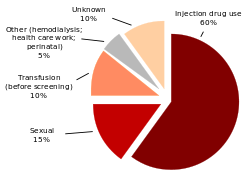
Hepatitis C is caused by the Hepatitis C virus (HCV).[3] In the scientific system that names and organizes viruses, the Hepatitis C virus is part of the hepacivirus genus in the family Flaviviridae.[9] There are seven major types of HCV, called "genotypes".[15] In the United States, the first genotype of HCV causes 70% of all cases of Hepatitis C ; the second genotype causes 20%; and each of the other genotypes causes 1% .[5] The first genotype is also the most common in South America and Europe.[3]
Transmission[change | change source]
In the developed world, the most common way that people get Hepatitis C is through intravenous drug use (by shooting drugs into a vein, using a needle that was already used by a person who has Hepatitis C). In the developing world, most people get Hepatitis C through blood transfusions (being given blood that was taken from a person with Hepatitis C) or by getting medical care with tools that were not cleaned enough after being used on a person with Hepatitis C.[16] In 20% of all cases of Hepatitis C, it is not known what caused the infection,[17] but many of these cases are thought to have been caused by intravenous drug use.[6]
Intravenous drug use[change | change source]
In many parts of the world, intravenous drug use is a major risk factor for Hepatitis C (meaning that it makes people more likely to get the disease).[18] One study that looked at 77 countries showed that in 25 of these countries (including the United States), between 60% and 80% of all IV drug users had Hepatitis C.[7] and China.[18] In twelve of the countries in the study, more than 80% of all IV drug users had Hepatitis C.[7] Across the world, as many as ten million IV drug users are thought to have Hepatitis C; the highest totals are in China (1.6 million), the United States (1.5 million), and Russia (1.3 million).[7] Studies have also shown that in places where there are high numbers of IV drug users, people are more likely to have Hepatitis C. Prisoners in the United States are ten to twenty times more likely than people in general to have Hepatitis C. This is because prisoners are more likely to do things that put them at high risk for getting Hepatitis C, like using IV drugs and getting tattoos with tools that have not been cleaned properly.[19][20]
Healthcare exposure[change | change source]
Peoples are at risk for getting Hepatitis C if they get blood transfusions (where a person is given blood from another person), blood products (which have blood or parts of blood in them), or organ transplants (where a person who needs a new organ is given an organ from another person), if these things have not been tested for the Hepatitis C virus.[5] In the United States, there has been universal screening - meaning that all blood and organs are tested before being given to another person - since 1992. Before then, about one out of every 200 units of blood carried the Hepatitis C virus;[21] since 1992, only one in 10,000 to 10,000,000 units of blood carry the virus.[6][17] The reason that there is still a low risk, instead of no risk at all, is that a person's blood does not test positive for the Hepatitis C virus until about 11–70 days after they get the disease.[17] So every once in a while, the screening tests may not pick up on a person's infection if the person got Hepatitis C less than 11–70 days before giving blood. While Hepatitis C screening works very well, some countries still do not screen blood and organ donations for the disease because of the cost.[11]
Sometimes, a healthcare worker will accidentally get stuck with a needle that was used on a person with Hepatitis C. If this happens, the healthcare worker has a small chance - about a 1.8% chance - of becoming infected.[5] The worker is more likely to get infected if the needle they were stuck with was hollow, or if the needle stuck deeply into their skin[11] It is also possible for the Hepatitis C virus to spread if an infected person's mucus touches another person's blood; however, the risk of this happening is low. The virus cannot be spread if an infected person's mucus touches another person's intact skin (skin that is whole and not damaged, with no wounds).[11]
Hepatitis C can also be spread through hospital equipment that has not been cleaned enough after being used on an infected person. Hepatitis C can be spread through needles, syringes, and medication containers that are re-used; through infusion bags (which are used to pump medications into a person's body; and through surgical equipment that is not sterile (or clean and free of germs).[11] In Egypt, which has the highest rate of infection in the world, medical and dental facilities with poor standards of care and cleanliness are the most common reason that Hepatitis C spreads.[22]
Sexual intercourse[change | change source]
Scientists do not know whether Hepatitis C can be spread through sex.[23] Hepatitis C is more likely in people who have sexual actions which make them much more likely to get Hepatitis C. However, it is not known whether this is because of these people's sexual behavior, or because these people were also using IV drugs.[5] There does not seem to be any risk that Hepatitis C can be spread through sexual contact between a heterosexual couple (a man and a woman; commonly called a "straight" couple) if neither person has sex with anyone else.[23] There does seem to be a risk of Hepatitis C spreading if one person already has a sexually transmitted infection, like HIV or genital ulceration; or if two people have sex in a way that causes wounds to the lining of the anal canal (like anal penetration - one person putting his penis into another person's anus).[23] The United States government says that most people need to use condoms to protect themselves from getting Hepatitis C only if they have more than one sexual partner.[24]
Body piercings[change | change source]
People who get tattoos are about two to three times more likely to get Hepatitis C.[25] This can be because of tools that are not clean or free of germs, or because the dyes used for tattooing are contaminated (the Hepatitis C virus has gotten inside of them).[25]
Tattoos or body piercings that were done before the mid-1980s or by people who are not professionals (not experts) are especially likely to spread Hepatitis C, since they are more likely to have used tools that were not sterile. Bigger tattoos also seem to put a person at more risk of getting Hepatitis C.[25] The risk of getting Hepatitis C is very high in prisons; in the United States. Almost half of all prison inmates share tattooing tools that are not sterile.[25] However, if a tattoo is done in a licensed place of business (which has to follow rules about cleaning tools and preventing diseases from spreading), there is almost no risk of getting Hepatitis C from the tattoo.[26]
Contact with blood[change | change source]
Because Hepatitis C is spread by blood-to-blood contact, personal-care tools which come into contact with blood - like razors, toothbrushes, and manicure or pedicure equipment or any other kind of blood to blood contact - can spread the disease if they are shared.[27][28] To prevent the spread of Hepatitis C, people should be careful with cuts, sores, or anything else that causes bleeding.[28] Hepatitis C does not spread through casual contact, such as hugging, kissing, or sharing eating or cooking utensils.[28]
Transmission from mother to child[change | change source]
Although this does not happen very often, a pregnant woman who has Hepatitis C can give the disease to her baby when it is born, or to her fetus while she is pregnant.[17] This happens in less than 10% of all pregnancies.[29] If a pregnant woman has Hepatitis C, nothing can be done to make her less likely to give her baby the disease.[29] If the woman is in labor (the process of delivering her baby) for a long time, there is more of a chance that the baby will become infected during the birth.[11] Breastfeeding does not seem to spread the disease. However, doctors say that an infected mother should not breastfeed if her nipples are cracked and bleeding,[30] or her viral loads (the amount of the Hepatitis C virus in her blood) are high.[17]
Diagnosis[change | change source]
There are a few different tests that can diagnose Hepatitis C (or say for sure that a person has the disease). These tests are called HCV antibody, ELISA, Western blot, and quantitative HCV RNA tests.[5] Polymerase chain reaction can show that a person has Hepatitis C virus RNA (part of the virus's genetic blueprint) one to two weeks after the person becomes infected.[12] Tests that look for Hepatitis C antibodies (which the body makes to fight the disease) cannot be done this quickly after infection, because the antibodies can take much longer to form and show up.[12]
A person has chronic Hepatitis C if he has been infected with the Hepatitis C virus for more than six months.[8] Because people with chronic Hepatitis C often have no symptoms for decades,[8] doctors often diagnose it through liver function tests (which test how well the liver is working) or by doing regular screening of high-risk people. Tests cannot tell whether an infection is acute or chronic.[11]
Blood testing[change | change source]
Hepatitis C testing usually begins with blood tests that look for antibodies to the Hepatitis C virus, using a technique called an enzyme immunoassay.[5] If this test is showing that the body is making antibodies to fight the Hepatitis C virus, the person will be tested a second time to make sure that the results are correct and to see how serious the infection is.[5] The results are confirmed using a technique called a recombinant immunoblot assay, and an HCV RNA polymerase chain reaction shows how serious the infection is.[5] If the immunoblot is positive, but there is no Hepatitis C virus RNA, this shows that the person had an infection, but it cleared up either with treatment or on its own. If the immunoblot is negative, this shows that the immunoassay (the first test) was wrong, and the person does not have Hepatitis C.[5] It takes six to eight weeks after a person gets infected before the immunoassay will test positive.[9]
During the first part of a Hepatitis C infection, a person's liver enzymes may change;[8] on average, they begin to rise seven weeks after infection.[9] A person's liver enzymes usually do not affect how severe their infection is.[9]
Biopsy[change | change source]
A test called a liver biopsy can show whether a person's liver has been damaged, or how badly it has been damaged, by Hepatitis C. There are some risks from the procedure.[3] In a biopsy, a doctor takes a small piece out of the patient's liver so that he can test it. There are three major changes to the liver that a biopsy usually shows. One is that lymphocytes (a type of white blood cell) show up in the liver tissue. Another is that lymphoid follicles (small masses or swellings) show up in the portal triad, a part of the liver. The third is changes to the bile ducts (these carry bile, which is made in the liver and is needed to help a person digest their food, to other parts of the body).[3] Many blood tests are available which try to measure how much damage there is to a patient's liver, in order to avoid the need for a biopsy and its risks.[3]
Screening[change | change source]
As few as 5–50% of infected people in the United States and Canada know that they have Hepatitis C.[25] Doctors say that people who are at high risk for the disease, including people with tattoos, should get tested.[25] Screening is also suggested for people with elevated (high) liver enzymes, since this is often the only sign that a person has chronic hepatitis.[31] Testing everyone is not recommended in the United States.[5]
Prevention[change | change source]
As of 2012, there is no vaccine that works to prevent Hepatitis C. Researchers are working on vaccines, and some are making progress.[32] The spread of Hepatitis C can be prevented by using a combination of strategies, like needle exchange programs, where IV drug users can get clean needles which will not spread the virus and treatment for drug abuse. If these strategies are used together, the risk of IV drug users getting Hepatitis C drops by about 75%.[33] Within each country screening blood donors is important. So is using universal precautions in healthcare facilities.[9] This means that healthcare workers treat every patient as if he has Hepatitis C, and they always wear gloves, clean their equipment properly, and keep things sterile so that diseases cannot be spread. In countries that do not have enough sterile syringes to use a new needle for every patient, healthcare providers should give medications by mouth rather than using a needle so that needles do not have to be re-used.[11]
Treatment[change | change source]
The Hepatitis C virus causes chronic infection in 50–80% of infected persons . About 40-80% of these cases clear up with treatment.[34][35] Although this almost never happens, chronic Hepatitis C can sometimes clear up on its own, without treatment.[6] Doctors say that people with chronic Hepatitis C should avoid drinking alcohol and taking medications that can be poisonous to the liver.[5] They also say that people with chronic infections should be given vaccines for Hepatitis A and Hepatitis B.[5] People with cirrhosis should also have ultrasound tests for liver cancer.[5]
Medications[change | change source]
If a person has changes in the liver that are not normal due to Hepatitis C virus infection, the person should get treatment.[5] The first treatment that is used is a drug called pegylated interferon given together with the virus-killing drug ribavirin. These medications are given for 24 or 48 weeks, depending on what type of Hepatitis C virus the person has.[5] About 50–60% of people who are treated improve.[5] For people with Hepatitis C virus genotype 1, treatment may work even better if another medication - either boceprevir or telaprevir - is given along with ribavirin and peginterferon alfa.[36][37][38] Side effects with treatment are common; half of people treated get flu-like symptoms, and a third have emotional problems.[5] Treatment works better if it is given during the first six months than after Hepatitis C becomes chronic.[12] If a person gets a new infection and it has not cleared up after eight to twelve weeks, doctors usually suggest giving pegylated interferon for another 24 weeks.[12] For people with thalassemia (a blood disorder), ribavirin appears to be a helpful treatment, but it makes it more likely for patients to need blood transfusions (where they need to be given blood from another person).[39] Some supporters of alternative medicine say that alternative therapies like milk thistle, ginseng, and colloidal silver can be helpful for Hepatitis C.[40] However, there is no proof that any alternative therapy has any effect on the Hepatitis C virus.[40][41][42]
Likely outcome[change | change source]
People respond differently to treatment, depending on which of the Hepatitis C virus genotypes they have. About 40-50% in people with genotype 1 have a good, stable response with 8–48 weeks of treatment.[3] In people with genotypes 2 and 3, about 70-80% have a good, stable response with 24 weeks of treatment.[3] About 65% of people with genotype 4 have a good, stable response with 48 weeks of treatment. There is not much evidence about how well treatment works for people with genotype 6 disease. The evidence that does exist looks at results after 48 weeks of treatment at the same medication doses as people with genotype 1.[43]
Epidemiology[change | change source]

no data <10 10-15 15-20 20-25 25-30 30-35 | 35-40 40-45 45-50 50-75 75–100 >100 |
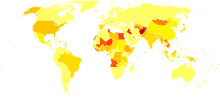
Between 130 and 170 million people, or about 3% of all of the people in the world, are living with chronic Hepatitis C.[44] Between 3–4 million people get infected per year. More than 350,000 people die every year from diseases caused by Hepatitis C.[44] The numbers of people getting Hepatitis C have increased a lot in the 20th century for a few different reasons. More people are using IV drugs. Also, more people are getting medical care with medical equipment that is not sterile, and it is more common for medications to be given intravenously.[11]
In the United States, about 2% of people have hepatitis C,[5] with 35,000 to 185,000 new cases a year. Rates have decreased in the West since the 1990s due to improved blood screening before transfusion.[12] Annual deaths from HCV in the United States range from 8,000 to 10,000. Expectations are that this mortality rate will increase as people infected by transfusion before HCV testing become ill and die.[45]
Infection rates are higher in some countries in Africa and Asia.[46] Countries with very high rates of infection include Egypt (22%), Pakistan (4.8%) and China (3.2%).[44] The high rate in Egypt is linked to a now-discontinued mass-treatment campaign for schistosomiasis, using improperly sterilized glass syringes.[11]
History[change | change source]
In the mid-1970s, Harvey J. Alter, Chief of the Infectious Disease Section in the Department of Transfusion Medicine at the National Institutes of Health, and his research team showed that most post-blood transfusion hepatitis cases were not due to hepatitis A or B viruses. Despite this discovery, international research efforts to identify the virus failed for the next decade. In 1987, Michael Houghton, Qui-Lim Choo, and George Kuo at Chiron Corporation, collaborating with Dr. D.W. Bradley from Centers for Disease Control and Prevention, used a new molecular cloning approach to identify the unknown organism and develop a diagnostic test.[47] In 1988, Alter confirmed the virus by verifying its presence in a panel of non A non B hepatitis specimens. In April 1989, the discovery of HCV was published in two articles in the journal Science.[48][49] The discovery led to significant improvements in diagnosis and improved antiviral treatment.[47] In 2000, Drs. Alter and Houghton were honored with the Lasker Award for Clinical Medical Research for "pioneering work leading to the discovery of the virus that causes hepatitis C and the development of screening methods that reduced the risk of blood transfusion-associated hepatitis in the U.S. from 30% in 1970 to virtually zero in 2000."[50]
Chiron filed for several patents on the virus and its diagnosis.[51] A competing patent application by the CDC was dropped in 1990 after Chiron paid $1.9 million to the CDC and $337,500 to Bradley. In 1994, Bradley sued Chiron, seeking to invalidate the patent, have himself included as a coinventor, and receive damages and royalty income. He dropped the suit in 1998 after losing before an appeals court.[52]
Society and culture[change | change source]
The World Hepatitis Alliance coordinates World Hepatitis Day, held every year on July 28.[53] The economic costs of hepatitis C are significant both to the individual and to society. In the United States the average lifetime cost of the disease was estimated at US$33,407 in 2003,[54] with the cost of a liver transplant approximately US$200,000 as of 2011.[55] In Canada the cost of a course of antiviral treatment was as high as 30,000 CAD in 2003,[56] while the United States costs are between 9,200 and 17,600 in 1998 USD.[54] In many areas of the world people are unable to afford treatment with antivirals because they lack insurance coverage or the insurance they have will not pay for antivirals.[57]
Research[change | change source]
As of 2011, about one hundred medications are in development for hepatitis C.[55] These medicines include vaccines to treat hepatitis, immunomodulators, and cyclophilin inhibitors.[58] These potentially new treatments have come about due to a better understanding of the hepatitis C virus.[59]
References[change | change source]
- ↑ 1.0 1.1 Ryan KJ, Ray CG, ed. (2004). Sherris Medical Microbiology (4th ed.). McGraw Hill. pp. 551–2. ISBN 0838585299.
- ↑ Houghton M (November 2009). "The long and winding road leading to the identification of the hepatitis C virus". Journal of Hepatology. 51 (5): 939–48. doi:10.1016/j.jhep.2009.08.004. PMID 19781804.
- ↑ 3.00 3.01 3.02 3.03 3.04 3.05 3.06 3.07 3.08 3.09 Rosen, HR (2011-06-23). "Clinical practice. Chronic hepatitis C infection" (PDF). The New England Journal of Medicine. 364 (25): 2429–38. doi:10.1056/NEJMcp1006613. PMID 21696309. Archived from the original (PDF) on 2012-12-15. Retrieved 2012-04-13.
- ↑ Maheshwari, A; Ray, S, Thuluvath, PJ (2008-07-26). "Acute hepatitis C.". Lancet. 372 (9635): 321–32. doi:10.1016/S0140-6736(08)61116-2. PMID 18657711. S2CID 46648044.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ↑ 5.00 5.01 5.02 5.03 5.04 5.05 5.06 5.07 5.08 5.09 5.10 5.11 5.12 5.13 5.14 5.15 5.16 5.17 5.18 5.19 5.20 5.21 Wilkins, T; Malcolm, JK, Raina, D. and Schade, RR (2010-06-01). "Hepatitis C: diagnosis and treatment". American Family Physician. 81 (11): 1351–7. PMID 20521755.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ↑ 6.0 6.1 6.2 6.3 6.4 Chronic Hepatitis C Virus Advances in Treatment, Promise for the Future. Springer Verlag. 2011. p. 4. ISBN 9781461411918.
- ↑ 7.0 7.1 7.2 7.3 Nelson, PK (2011-08-13). "Global epidemiology of hepatitis B and hepatitis C in people who inject drugs: results of systematic reviews". Lancet. 378 (9791). Mathers, BM, Cowie, B, Hagan, H, Des Jarlais, D, Horyniak, D, Degenhardt, L: 571–83. doi:10.1016/S0140-6736(11)61097-0. PMC 3285467. PMID 21802134.
- ↑ 8.0 8.1 8.2 8.3 Chronic Hepatitis C Virus Advances in Treatment, Promise for the Future. Springer Verlag. 2011. pp. 103–104. ISBN 9781461411918.
- ↑ 9.0 9.1 9.2 9.3 9.4 9.5 Gerald L. Mandell, John E. Bennett, Raphael Dolin, ed. (2010). Mandell, Douglas, and Bennett's principles and practice of infectious diseases (7th ed.). Philadelphia, PA: Churchill Livingstone/Elsevier. pp. Chapter 154. ISBN 978-0443068393.
{{cite book}}: CS1 maint: multiple names: editors list (link) - ↑ Mueller, S; Millonig, G. and Seitz, HK (2009-07-28). "Alcoholic liver disease and hepatitis C: a frequently underestimated combination". World Journal of Gastroenterology : WJG. 15 (28): 3462–71. doi:10.3748/wjg.15.3462. PMC 2715970. PMID 19630099.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ↑ 11.00 11.01 11.02 11.03 11.04 11.05 11.06 11.07 11.08 11.09 Alter, MJ (2007-05-07). "Epidemiology of hepatitis C virus infection". World Journal of Gastroenterology : WJG. 13 (17): 2436–41. doi:10.3748/wjg.v13.i17.2436 (inactive 2024-01-24). PMC 4146761. PMID 17552026.
{{cite journal}}: CS1 maint: DOI inactive as of January 2024 (link) - ↑ 12.0 12.1 12.2 12.3 12.4 12.5 Ozaras, R; Tahan, V (Apr 2009). "Acute hepatitis C: prevention and treatment". Expert Review of Anti-infective Therapy. 7 (3): 351–61. doi:10.1586/eri.09.8. PMID 19344247. S2CID 25574917.
- ↑ Zignego AL, Ferri C, Pileri SA, Caini P, Bianchi FB (January 2007). "Extrahepatic manifestations of Hepatitis C Virus infection: a general overview and guidelines for a clinical approach". Digestive and Liver Disease. 39 (1): 2–17. doi:10.1016/j.dld.2006.06.008. PMID 16884964.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ↑ Louie, KS; Micallef, JM, Pimenta, JM and Forssen, UM (Jan 2011). "Prevalence of thrombocytopenia among patients with chronic hepatitis C: a systematic review". Journal of Viral Hepatitis. 18 (1): 1–7. doi:10.1111/j.1365-2893.2010.01366.x. PMID 20796208. S2CID 21412295.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ↑ Nakano T, Lau GM, Lau GM, Sugiyama M, Mizokami M (December 2011). "An updated analysis of hepatitis C virus genotypes and subtypes based on the complete coding region". Liver Int. 32 (2): 339–345. doi:10.1111/j.1478-3231.2011.02684.x. PMID 22142261. S2CID 23271017.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ↑ Maheshwari, A; Thuluvath, PJ (Feb 2010). "Management of acute hepatitis C.". Clinics in Liver Disease. 14 (1): 169–76, x. doi:10.1016/j.cld.2009.11.007. PMID 20123448.
- ↑ 17.0 17.1 17.2 17.3 17.4 Pondé, RA; Mikhaĭlova, A (Feb 2011). "Hidden hazards of HCV transmission". Medical Microbiology and Immunology. 200 (1): 7–11. doi:10.1007/s00430-010-0159-9. PMID 20461405. S2CID 664199.
- ↑ 18.0 18.1 Xia, X; Luo, J, Bai, J, Yu, R (Oct 2008). "Epidemiology of HCV infection among injection drug users in China: systematic review and meta-analysis". Public Health. 122 (10): 990–1003. doi:10.1016/j.puhe.2008.01.014. PMID 18486955.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ↑ Imperial, JC (Jun 2010). "Chronic hepatitis C in the state prison system: insights into the problems and possible solutions". Expert Review of Gastroenterology & Hepatology. 4 (3): 355–64. doi:10.1586/egh.10.26. PMID 20528122. S2CID 7931472.
- ↑ Vescio, MF (Apr 2008). "Correlates of hepatitis C virus seropositivity in prison inmates: a meta-analysis". Journal of Epidemiology and Community Health. 62 (4). Longo, B, Babudieri, S, Starnini, G, Carbonara, S, Rezza, G, Monarca, R: 305–13. doi:10.1136/jech.2006.051599. PMID 18339822. S2CID 206989111.
- ↑ Marx, John (2010). Rosen's emergency medicine: concepts and clinical practice 7th edition. Philadelphia, PA: Mosby/Elsevier. p. 1154. ISBN 9780323054720.
- ↑ "Highest Rates of Hepatitis C Virus Transmission Found in Egypt". Al Bawaba. 2010-08-09. Archived from the original on 2012-05-15. Retrieved 2010-08-27.
- ↑ 23.0 23.1 23.2 Tohme RA, Holmberg SD (June 2010). "Is sexual contact a major mode of hepatitis C virus transmission?". Hepatology. 52 (4): 1497–505. doi:10.1002/hep.23808. PMID 20635398. S2CID 5592006.
- ↑ "Hepatitis C Group Education Class". United States Department of Veteran Affairs.
- ↑ 25.0 25.1 25.2 25.3 25.4 25.5 Jafari, S (Nov 2010). "Tattooing and the risk of transmission of hepatitis C: a systematic review and meta-analysis" (PDF). International Journal of Infectious Diseases : IJID : Official Publication of the International Society for Infectious Diseases. 14 (11). Copes, R, Baharlou, S, Etminan, M, Buxton, J: e928-40. doi:10.1016/j.ijid.2010.03.019. PMID 20678951.
- ↑ "Hepatitis C" (PDF). Center for Disease Control and Prevention. Retrieved 2 January 2012.
- ↑ Lock G, Dirscherl M, Obermeier F; et al. (September 2006). "Hepatitis C —contamination of toothbrushes: myth or reality?". J. Viral Hepat. 13 (9): 571–3. doi:10.1111/j.1365-2893.2006.00735.x. PMID 16907842. S2CID 24264376.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ↑ 28.0 28.1 28.2 "Hepatitis C". FAQ – CDC Viral Hepatitis. Retrieved 2 January 2012.
- ↑ 29.0 29.1 Lam, NC; Gotsch, PB and Langan, RC (2010-11-15). "Caring for pregnant women and newborns with hepatitis B or C.". American Family Physician. 82 (10): 1225–9. PMID 21121533.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ↑ Mast EE (2004). "Mother-to-Infant Hepatitis C Virus Transmission and Breastfeeding". Protecting Infants through Human Milk. Advances in Experimental Medicine and Biology. Vol. 554. pp. 211–6. doi:10.1007/978-1-4757-4242-8_18. ISBN 978-1-4419-3461-1. PMID 15384578.
- ↑ Senadhi, V (Jul 2011). "A paradigm shift in the outpatient approach to liver function tests". Southern Medical Journal. 104 (7): 521–5. doi:10.1097/SMJ.0b013e31821e8ff5. PMID 21886053. S2CID 26462106.
- ↑ Halliday, J; Klenerman, P. and Barnes, E (May 2011). "Vaccination for hepatitis C virus: closing in on an evasive target". Expert Review of Vaccines. 10 (5): 659–72. doi:10.1586/erv.11.55. PMC 3112461. PMID 21604986.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ↑ Hagan, H; Pouget, ER and Des Jarlais, DC (2011-07-01). "A systematic review and meta-analysis of interventions to prevent hepatitis C virus infection in people who inject drugs". The Journal of Infectious Diseases. 204 (1): 74–83. doi:10.1093/infdis/jir196. PMC 3105033. PMID 21628661.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ↑ Torresi, J; Johnson, D. and Wedemeyer, H (Jun 2011). "Progress in the development of preventive and therapeutic vaccines for hepatitis C virus". Journal of Hepatology. 54 (6): 1273–85. doi:10.1016/j.jhep.2010.09.040. PMID 21236312.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ↑ Ilyas, JA; Vierling, JM (Aug 2011). "An overview of emerging therapies for the treatment of chronic hepatitis C.". Clinics in Liver Disease. 15 (3): 515–36. doi:10.1016/j.cld.2011.05.002. PMID 21867934.
- ↑ Foote BS, Spooner LM, Belliveau PP (September 2011). "Boceprevir: a protease inhibitor for the treatment of chronic hepatitis C". Ann Pharmacother. 45 (9): 1085–93. doi:10.1345/aph.1P744. PMID 21828346. S2CID 39593200.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ↑ Smith LS, Nelson M, Naik S, Woten J (May 2011). "Telaprevir: an NS3/4A protease inhibitor for the treatment of chronic hepatitis C". Ann Pharmacother. 45 (5): 639–48. doi:10.1345/aph.1P430. PMID 21558488. S2CID 2886659.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ↑ Ghany MG, Nelson DR, Strader DB, Thomas DL, Seeff LB (October 2011). "An update on treatment of genotype 1 chronic hepatitis C virus infection: 2011 practice guideline by the American Association for the Study of Liver Diseases". Hepatology. 54 (4): 1433–44. doi:10.1002/hep.24641. PMC 3229841. PMID 21898493.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ↑ Alavian SM, Tabatabaei SV (April 2010). "Treatment of chronic hepatitis C in polytransfused thalassaemic patients: a meta-analysis". J. Viral Hepat. 17 (4): 236–44. doi:10.1111/j.1365-2893.2009.01170.x. PMID 19638104. S2CID 20271240.
- ↑ 40.0 40.1 Hepatitis C and CAM: What the Science Says Archived 2013-05-13 at the Wayback Machine. NCCAM March 2011. (Retrieved 07 March 2011)
- ↑ Liu, J; Manheimer, E. and Tsutani, K. and Gluud, C (Mar 2003). "Medicinal herbs for hepatitis C virus infection: a Cochrane hepatobiliary systematic review of randomized trials". The American Journal of Gastroenterology. 98 (3): 538–44. doi:10.1111/j.1572-0241.2003.07298.x. PMID 12650784. S2CID 20014583.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ↑ Rambaldi, A; Jacobs, BP, Gluud, C (2007-10-17). "Milk thistle for alcoholic and/or hepatitis B or C virus liver diseases". Cochrane Database of Systematic Reviews (Online). 2009 (4): CD003620. doi:10.1002/14651858.CD003620.pub3. PMC 8724782. PMID 17943794.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ↑ Fung J, Lai CL, Hung I; et al. (September 2008). "Chronic hepatitis C virus genotype 6 infection: response to pegylated interferon and ribavirin". The Journal of Infectious Diseases. 198 (6): 808–12. doi:10.1086/591252. PMID 18657036. S2CID 22715733.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ↑ 44.0 44.1 44.2 "WHO Hepatitis C factsheet". 2011. Retrieved 2011-07-13.
- ↑ J. M. Colacino; Heinz, B. A., eds. (2004). Hepatitis prevention and treatment. Basel: Birkhäuser. p. 32. ISBN 9783764359560.
- ↑ Gary W. Brunette; et al., eds. (12 May 2011). CDC health information for international travel : the Yellow Book 2012. New York: Oxford University. p. 231. ISBN 9780199769018.
- ↑ 47.0 47.1 Boyer, JL (2001). Liver cirrhosis and its development: proceedings of the Falk Symposium 115. Springer. pp. 344. ISBN 9780792387602.
- ↑ Choo QL, Kuo G, Weiner AJ, Overby LR, Bradley DW, Houghton M (April 1989). "Isolation of a cDNA clone derived from a blood-borne non-A, non-B viral hepatitis genome". Science. 244 (4902): 359–62. Bibcode:1989Sci...244..359C. doi:10.1126/science.2523562. PMID 2523562.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ↑ Kuo G, Choo QL, Alter HJ; et al. (April 1989). "An assay for circulating antibodies to a major etiologic virus of human non-A, non-B hepatitis". Science. 244 (4902): 362–4. Bibcode:1989Sci...244..362K. doi:10.1126/science.2496467. PMID 2496467.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ↑ Winners Albert Lasker Award for Clinical Medical Research, The Lasker Foundation. Retrieved 20 February 2008.
- ↑ Houghton, M., Q.-L. Choo, and G. Kuo. NANBV Diagnostics and Vaccines. European Patent No. EP-0-3 18-216-A1. European Patent Office (filed 18 November 1988, published 31 May 1989).
- ↑ Wilken, Judge. "United States Court of Appeals for the Federal Circuit". Retrieved 11 January 2012.
- ↑ Eurosurveillance editorial, team (2011-07-28). "World Hepatitis Day 2011". Euro Surveillance : Bulletin Europeen Sur les Maladies Transmissibles = European Communicable Disease Bulletin. 16 (30). PMID 21813077.
- ↑ 54.0 54.1 Wong, JB (2006). "Hepatitis C: cost of illness and considerations for the economic evaluation of antiviral therapies". PharmacoEconomics. 24 (7): 661–72. doi:10.2165/00019053-200624070-00005. PMID 16802842. S2CID 6713508.
- ↑ 55.0 55.1 El Khoury, A.C.; Klimack, W.K.; Wallace, C.; Razavi, H. (1 December 2011). "Economic burden of hepatitis C-associated diseases in the United States". Journal of Viral Hepatitis. 19 (3): 153–160. doi:10.1111/j.1365-2893.2011.01563.x. PMID 22329369. S2CID 27409621.
- ↑ "Hepatitis C Prevention, Support and Research ProgramHealth Canada". Public Health Agency of Canada. Nov 2003. Retrieved 10 January 2012.
- ↑ Howard Thomas, Stanley Lemon, Arie Zuckerman, ed. (2008). Viral Hepatitis (3rd ed.). Oxford: John Wiley & Sons. p. 532. ISBN 9781405143882.
{{cite book}}: CS1 maint: multiple names: editors list (link) - ↑ Ahn, J; Flamm, SL (Aug 2011). "Hepatitis C therapy: other players in the game". Clinics in Liver Disease. 15 (3): 641–56. doi:10.1016/j.cld.2011.05.008. PMID 21867942.
- ↑ Vermehren, J; Sarrazin, C (Feb 2011). "New HCV therapies on the horizon". Clinical Microbiology and Infection : The Official Publication of the European Society of Clinical Microbiology and Infectious Diseases. 17 (2): 122–34. doi:10.1111/j.1469-0691.2010.03430.x. PMID 21087349. S2CID 32983889.
