Severe acute respiratory syndrome coronavirus 2
| Severe acute respiratory syndrome coronavirus 2 | |
|---|---|

| |
| Transmission electron micrograph of SARS-CoV-2 virions with visible coronae | |
| |
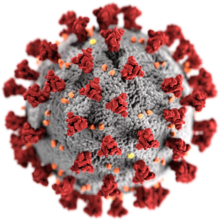
| Illustration of a SARS-CoV-2 virion[1] Grey coating: the envelope, composed mainly of lipids, which can be destroyed with alcohol or soap[1] Yellow deposits: envelope proteins (E)[1]
| |
| Virus classification | |
| (unranked): | Virus |
| Realm: | incertae sedis |
| Kingdom: | incertae sedis |
| Phylum: | incertae sedis |
| Class: | incertae sedis |
| Order: | Nidovirales |
| Family: | Coronaviridae |
| Genus: | Betacoronavirus |
| Subgenus: | Sarbecovirus |
| Species: | |
| Strain: | Severe acute respiratory syndrome coronavirus 2
|
| Synonyms | |
| |
SARS coronavirus 2 (SARS-CoV-2) is a positive-sense, single-stranded RNA coronavirus which causes the disease COVID-19.[2] It used to be known as 2019 novel coronavirus (2019-nCoV) by the World Health Organization (WHO).[3][4]
The virus started the 2019–20 coronavirus outbreak.[5][6][7] The first suspected cases were reported to the WHO on December 31, 2019.[8][9] Many early cases of this new coronavirus were linked to Huanan Seafood Wholesale Market, a large seafood and animal market in Wuhan, China. The virus may have come from infected animals. It is not certain that this place was the source of the pandemic.[10]
Origins[change | change source]
The genetic material of this virus showed many similarities to SARS-CoV (79.5%)[11] and bat coronaviruses (96%).[11] This means the virus may have originally come from bats.[12][13][14] Scientists did more experiments that showed the virus probably went from bats to an intermediate host, meaning another animal in between bats and humans. The viruses in that other animal changed over time until they could infect humans. Scientists are close to sure that the original animal was a bat but not sure what the intermediate animal was.[15] Some scientists think it could have been a pangolin because there are coronaviruses that live in pangolins even though they are not exactly the same as SARS-CoV-2 or the ones in bats.[16] Pangolins are an endangered species and buying, selling or moving them from place to place is illegal in China and many other countries. But their scales are an ingredient in many traditional Chinese medicines, so they are often sold on the black market.[17]
Although SARS-CoV-2 began in bats, scientists noticed that the bats do not get sick and die from it. They think this is because the bats have very careful immune systems that fight the virus without causing inflammation. Scientists are studying bats because of this.[18][19]
Scientists are still looking at SARS-CoV-2 infection in animals, but only a variant strain has been confirmed to spread to minks.[20] It is also inducing fear in humans as there are cases being reported of transmission of COVID-19 from minks to humans.[21] Cats, dogs and other mammals are said to at relative risk but how they can get infected is still a mystery.[22]
Name[change | change source]

In early February 2020, the International Committee on the Taxonomy of Viruses' Coronavirus Study Group gave SARS-CoV-2 its official name, for sudden acute respiratory syndrome coronavirus number two. Before that, people called the virus "2019-nCoV," for "novel (new) coronavirus that appeared in 2019." The study group chose this name because the new virus was so similar to another virus that was already called SARS-CoV with no number.[23][24]
How the virus causes disease[change | change source]
First, the virus enters the cell with the help of the S protein or spike protein expressed on the surface of the virus beyond its envelope.[25] The spike protein attaches to the human cell receptor hACE2 or human angiotensin-converting enzyme 2 . ACE2 is found on the cells of the epithelial cell linings of the lungs, kidneys, and intestines.[26] After the attachment of the S- protein to the virus, the S protein splits into its two components- S1 head and S2 stalk . The S1 head is now able to bind to the TMPRSS2 receptor which is a serine protease enzyme containing a transmembrane domain. The interaction of TMPRSS2 and S1 trigger the cell membrane of the human cell to merge with that of the virus leading to viral genome entry into cells via endosomes.[27][28] The viral genome in the endosomes which convert to lysosomes is then processed by cathepsin, which is then replicated in the human cells and released after viral packaging and maturation in the cells to infect others. In October 2020, scientists said they had seen SARS-CoV-2 use another receptor, Neuropilin-1, to enter cells with the help of the furin cleavage site in its genome which was not found in any of its previous ancestors.[27][29][30]
SARS-CoV-2 can infect many kinds of cells, but it mostly infects cells in the respiratory system, such as the cells of the nose, pharynx and lungs. Because it is in cells inside the nose, it can spread via aerosol or droplets when people breathe, talk, cough or sneeze.[30]
The expanding part of the lungs, pulmonary alveoli, have two main types of cells. One cell, type I, absorbs oxygen from the air, i.e. gas exchange. The other, type II, produces surfactants, that help keep the lungs fluid, clean, infection free, etc. COVID-19 finds a way into a surfactant producing type II cell, and smothers it by reproducing COVID-19 virus within it. Each type II cell which is killed by the virus causes an extreme reaction in the lungs. Fluids, pus, and dead cell material flood the lung, causing the coronavirus pulmonary disease.[31]
Lung damage[change | change source]
Scientists looked at the lungs from people who died of COVID-19. They compared them to lungs from people who died of influenza A and to lungs from people who died but not from any problem with their lungs. They saw the cells that made up the skins of the blood vessels in the lungs were more badly damaged in the lungs from COVID-19 patients, and there was more blood clotting. The most important difference the scientists saw was that the lungs had begun to grow new blood vessels.[32][33]
COVID-19[change | change source]
In February 2020, the World Health Organization announced they had chosen a name for the disease caused by SARS-CoV-2: COVID-19. "Covi" for "coronavirus," "D" for "disease," and "19" for the year 2019. They said they did not want the name to have any person, place, or animal in it, like "Wuhan," or “pangolin,” because then people might blame the disease on that place, person, or animal. They also wanted the name to be easy to say out loud.[2]
Symptoms[change | change source]
According to the United States Centers for Disease Control and Prevention, COVID-19 makes people feel sick in different ways, but it usually affects the lungs. People usually cough and have difficulty breathing. They often also have a fever, chills, headache, pain in their muscles, or trouble tasting or smelling things.[34]
According to an April 2020 study by the American Gastroenterological Association, COVID-19 can make sick people vomit or have diarrhea but this is rare. They said about 7.7% of COVID-19 patients threw up, about 7.8% had diarrhea and about 3.6% had pain in their stomachs.[35]
Painkilling[change | change source]
A study from the University of Arizona found that SARS-CoV-2 can prevent an infected person from feeling pain. It does this by giving off vascular endothelial growth factor-A, which binds to the human body's neuropilin receptor. This way, sick people do not feel pain, do not know they are sick, and spread the virus more than if they could feel pain.[36]
Shape[change | change source]
The SARS-CoV-2 virus looks like a round ball with spikes all around it. There are four parts to the virus: the spikes, a membrane, an envelope, and the virus' genetic material, or ribonucleic acid (RNA) Each of these four parts is a different protein molecule. The spikes, membrane, and envelope are together called the viral envelope, or outer layer, of the virus.[37][38]
SARS-CoV-2 is a positive-strand RNA virus, meaning that it uses ribonucleic acid (RNA) to hold the patterns to make the proteins it needs instead of using DNA the way humans and other multicellular living things do.[39]
Conspiracy theories[change | change source]
In early 2020, some people began to think that the SARS-CoV-2 may have been made on purpose in a laboratory at the Wuhan Institute of Virology and released in Wuhan like a weapon. When the leader of Iran, Ayatollah Khamenei, said that he did not want the United States to help his country against coronavirus, he named the idea that Americans had made the virus on purpose to harm Iranains as one of his reasons: "I do not know how real this accusation is but when it exists, who in their right mind would trust you to bring them medication?" said Khamenei.[40]
One survey by Pew Research showed 29% of Americans who answered, thought SARS-CoV-2 could have been made in a lab on purpose and 23% thought it could have been made in a lab by accident.[41] A survey of people in the United Kingdom showed many of them thought COVID-19 was caused by 5G wireless networks.[42]
On March 17, 2020, scientists from Columbia University and other places published a paper in Nature Medicine showing that SARS-CoV-2 was almost surely not made by humans in a laboratory. They did this by comparing the genomes of different viruses to each other.[16] The scientists saw that SARS-CoV-2 did not match any of the viral backbones that already exist for virologists to use.[43] The paper in Nature Medicine soon became one of the most cited scientific papers of all time.[16]
Medicines[change | change source]
There are no fixed cures for the treatment of COVID-19, but there are various drugs that have been approved for use such as Hydroxychloroquine and Remdesivir which are detailed below. Other antiviral drugs, interferon therapy and combination of anti-viral and interferons are also being experimented on to get the best possible outcome for patients. These treatments are used to reduce the symptoms and to keep the patients comfortable.[44][45]
In April 2020, scientists from the University of Pittsburgh said they had made a vaccine, called PittCoVacc and tested it in mice.[46][47]
Another team of scientists led by Dr. Josef Penninger of the University of British Columbia invented a medicine called APN01 and tested it in engineered human tissue, meaning human cells put together in a lab to look and act like they were inside a body. The scientists learned that adding human soluble recombinant angiotensin converting enzyme 2 (ACE2) to these tissues infected with SARS-CoV-2 made it harder for the virus to reproduce.[48][49]
In late April 2020, a team from Oxford University announced that they had developed a COVID-19 vaccine. The United States National Institutes of health tested it in rhesus monkeys, and it worked. The Oxford scientists said because they had already been working on a vaccine against a different coronavirus, they had a head start working on one for SARS-CoV-2. The scientists said they would try to test their vaccine on 6000 people by the end of May 2020, and that their vaccine could be ready for people to use in September 2020.[50][51]
In mid-May 2020, a company called Moderna said they tested their mRNA vaccine in forty-five people and eight of them produced antibodies[52] but they did not publish the specific data or publish an article in a scientific journal.[53] Anna Durbin of Johns Hopkins University said it was too soon to tell if people would keep the antibodies long enough for the vaccine to work.[54] The United States Food and Drug Administration gave Moderna permission to test the vaccine again in more people. Moderna's chief medical officer said the vaccine could be ready in January 2021.[52]
Hydroxychloroquine[change | change source]
Some people think hydroxychloroquine, a medicine given to people with malaria, lupus, and arthritis, could work against COVID-19 and some do not. One study from China showed that COVID-19 patients who took hydroxychloroquine got better faster, but the study was not peer reviewed. Other studies in France and China seemed to show hydroxychloroquine helped, but the studies did not include control groups, meaning the doctors did not compare patients who took hydroxychloroquine to patients who did not, so they could not be sure it was the hydroxychloroquine that was helping them or whether it was something else.[55] In March, the United States Food and Drug Administration gave doctors permission to give hydroxychloroquine to COVID-19 patients.[56]
United States President Donald Trump suggested the malaria drug hydroxychloroquine could help cure COVID-19, but Dr. Anthony Fauci, who is part of the White House's official coronavirus team said no one could know if hydroxychloroquine worked against SARS-CoV-2. In early April, the New York Times reported that President Trump has "a small personal financial interest" in Sanofi, one of the companies that makes hydroxychloroquine, meaning that if the company sold more hydroxychloroquine, he would have more wealth.[56]
In early April, Fauci said, "The data are really just at best suggestive. There have been cases that show there may be an effect and there are others to show there's no effect."[57] Dr. Megan L. Ranney of Brown University said that hydroxychloroquine can cause heart attacks and other problems. Other doctors worry that if people take hydroxychloroquine for COVID-19, there will not be enough left for people with malaria, lupus, and arthritis. Still, some hospitals have given hydroxychloroquine to COVID-19 patients who are very sick because the doctors think it is worth the risk.[55]
Scientists in France and China performed more studies on larger groups of patients taking hydroxychloroquine. They watched patients who were taking the medicine and other treatment together and at patients who were only taking the other treatment. Both studies showed that hydroxychloroquine did not help and did cause side effects. Both studies were published in May 2020.[58]
Remdesivir[change | change source]
Some scientists thought the drug remdesivir, which was invented as a medicine for Ebola, could work against SARS-CoV-2. Remdesivir works against some viruses. Remdesivir had already been tested in humans, so the doctors already knew it would not hurt the patients even if it did not make them better. Because scientists already knew remdesivir was safe, they were able to start testing it in humans right away.[59][60]
In one study, doctors gave a drug called remdesivir to 61 COVID-19 patients on a compassionate basis, meaning they gave them the drug because there was no other treatment available. Scientists studied 53 of these patients and found that 68% of the patients got better, 13% died, and 25% had serious side effects. But because the study had no control group, meaning these patients were not compared to other COVID-19 patients who were not taking remdesivir, and because only 53 people were in the experiment, scientists must run more studies before they can be sure remdesivir works.[59][60]
The chairman and CEO of the company that makes remdesivir, David O'Day, said that remdesivir might work better in some patients than in others and asked scientists to perform many different kinds of studies. [59][60]
In October 2020, the National Institutes of Health told the world the results of its tests: They studied 11,000 people and found that remdesivir did not help keep COVID-19 patients alive.[61]
Related pages[change | change source]
References[change | change source]
- ↑ 1.0 1.1 1.2 1.3 1.4 Giaimo C (1 April 2020). "The Spiky Blob Seen Around the World". The New York Times. Archived from the original on 2 April 2020. Retrieved 6 April 2020.
- ↑ 2.0 2.1 Sanya Mansoor (February 11, 2020). "What's in a Name? Why WHO's Formal Name for the New Coronavirus Disease Matters". Time. Retrieved February 13, 2020.
- ↑ "Surveillance case definitions for human infection with novel coronavirus (nCoV)". who.int. Retrieved 21 January 2020.
- ↑ "Novel coronavirus (2019-nCoV), Wuhan, China". cdc.gov. 10 January 2020. Retrieved 16 January 2020.
- ↑ "中国疾病预防控制中心". chinacdc.cn. Retrieved 9 January 2020.
- ↑ "New-type coronavirus causes pneumonia in Wuhan: expert". Xinhua. Archived from the original on 9 January 2020. Retrieved 9 January 2020.
- ↑ "CoV2020". platform.gisaid.org. Retrieved 12 January 2020.
- ↑ "Pneumonia of unknown cause – China. Disease outbreak news". World Health Organization. 5 January 2020. Archived from the original on 7 January 2020. Retrieved 6 January 2020.
- ↑ Schnirring, Lisa (14 January 2020). "Report: Thailand's coronavirus patient didn't visit outbreak market". CIDRAP. Archived from the original on 14 January 2020. Retrieved 15 January 2020.
- ↑ "Update and Interim Guidance on Outbreak of 2019 Novel Coronavirus (2019-nCoV) in Wuhan, China CDC Health Update". New Jersey Department of Health. 18 January 2020
- ↑ 11.0 11.1 Zhou, Peng; Yang, Xing-Lou; Wang, Xian-Guang; Hu, Ben; Zhang, Lei; Zhang, Wei; Si, Hao-Rui (23 January 2020). "Discovery of a novel coronavirus associated with the recent pneumonia outbreak in humans and its potential bat origin". bioRxiv: 2020.01.22.914952. doi:10.1101/2020.01.22.914952. S2CID 211003249 – via www.biorxiv.org.
- ↑ Sample CoVZC45 and CoVZXC21, see there for an interactive visualisation Archived 20 January 2020 at the Wayback Machine
- ↑ Benvenuto, Domenico; Giovannetti, Marta; Ciccozzi, Alessandra; Spoto, Silvia; Angeletti, Silvia; Ciccozzi, Massimo (2020). "The 2019 new Coronavirus epidemic: evidence for virus evolution". bioRxiv. 92 (4): 455–459. doi:10.1101/2020.01.24.915157. PMC 7166400. PMID 31994738.
- ↑ Callaway, Ewen; Cyranoski, David (23 January 2020). "Why snakes probably aren't spreading the new China virus". Nature. doi:10.1038/d41586-020-00180-8. PMID 33479530. S2CID 214391572. Archived from the original on 25 January 2020. Retrieved 27 January 2020.
- ↑ James Gorman (March 26, 2020). "Significance of Pangolin Viruses in Human Pandemic Remains Murky: Scientists haven't found evidence that the new coronavirus jumped from pangolins to people, but they do host very similar viruses". New York Times. Retrieved March 31, 2020.
- ↑ 16.0 16.1 16.2 *University of Sydney (March 26, 2020). "Unlocking the Genetic Code of the Novel Coronavirus: How COVID-19 Made the Leap From Animals to Humans". SciTech Daily. Retrieved March 29, 2020.
- ↑ *Charlie Campbell and Mong La (November 21, 2016). "Traditional Chinese Medical Authorities Are Unable to Stop the Booming Trade in Rare Animal Parts". Time.
- ↑ "Bats offer clues to treating COVID-19" (Press release). University of Rochester. July 8, 2020. Retrieved July 11, 2020.
- ↑ Vera Gorbunova; Andrei Seluanov; Brian K. Kennedy (July 7, 2020). "The World Goes Bats:Living Longer and Tolerating Viruses" (PDF). Cell Metabolism. 32 (1). Cell Press: 31–43. doi:10.1016/j.cmet.2020.06.013. PMC 7341951. PMID 32640245. Retrieved July 11, 2020.
- ↑ "What is it about mink and COVID-19?". Newsletters. 2020-08-20. Retrieved 2021-03-06.
- ↑ Doheny, Kathleen. "COVID in Minks Spreading to Humans". WebMD. Retrieved 2021-03-06.
- ↑ CDC (2020-02-11). "COVID-19 and Your Health". Centers for Disease Control and Prevention. Retrieved 2021-03-06.
- ↑ Alexander E. Gorbalenya; Susan C. Baker; Ralph S. Baric; Raoul J. de Groot; Christian Drosten; Anastasia A. Gulyaeva; Bart L. Haagmans; Chris Lauber; Andrey M Leontovich; Benjamin W. Neuman; Dmitry Penzar; Stanley Perlman; Leo L.M. Poon; Dmitry Samborskiy; Igor A. Sidorov; Isabel Sola11; John Ziebuhr (February 11, 2020). "Severe acute respiratory syndrome-related coronavirus – The species and its viruses, a statement of the Coronavirus Study Group". International Committee on the Taxonomy of Viruses. doi:10.1101/2020.02.07.937862. S2CID 214317727. Retrieved April 17, 2020.
{{cite journal}}: Cite journal requires|journal=(help)CS1 maint: numeric names: authors list (link) - ↑ Yuntao Wu; Wenzhe Ho; Yaowei Huang; Dong-Yan Jin; Shiyue Li; Shan-Lu Liu; Xuefeng Liu; Jianming Qiu; Yongming Sang; Qiuhong Wang; Kwok-Yung Yuen; Zhi-Ming Zheng (March 6, 2020). "SARS-CoV-2 is an appropriate name for the new coronavirus". Lancet. 395 (10228): 949–950. doi:10.1016/S0140-6736(20)30557-2. PMC 7133598. PMID 32151324. S2CID 212423659.
- ↑ Huang, Yuan; Yang, Chan; Xu, Xin-feng; Xu, Wei; Liu, Shu-wen (September 2020). "Structural and functional properties of SARS-CoV-2 spike protein: potential antivirus drug development for COVID-19". Acta Pharmacologica Sinica. 41 (9): 1141–1149. doi:10.1038/s41401-020-0485-4. ISSN 1745-7254. PMC 7396720. PMID 32747721.
- ↑ "Angiotensin-converting enzyme 2". Wikipedia. 2021-02-24.
- ↑ 27.0 27.1 Shang, Jian; Wan, Yushun; Luo, Chuming; Ye, Gang; Geng, Qibin; Auerbach, Ashley; Li, Fang (2020-05-26). "Cell entry mechanisms of SARS-CoV-2". Proceedings of the National Academy of Sciences. 117 (21): 11727–11734. doi:10.1073/pnas.2003138117. ISSN 0027-8424. PMC 7260975. PMID 32376634.
- ↑ Wu, Canrong; Liu, Yang; Yang, Yueying; Zhang, Peng; Zhong, Wu; Wang, Yali; Wang, Qiqi; Xu, Yang; Li, Mingxue (2020-02-27). "Analysis of therapeutic targets for SARS-CoV-2 and discovery of potential drugs by computational methods". Acta Pharmaceutica Sinica. B. 10 (5): 766–788. doi:10.1016/j.apsb.2020.02.008. ISSN 2211-3835. PMC 7102550. PMID 32292689.
- ↑ Cantuti-Castelvetri, Ludovico; Ojha, Ravi; Pedro, Liliana D.; Djannatian, Minou; Franz, Jonas; Kuivanen, Suvi; van der Meer, Franziska; Kallio, Katri; Kaya, Tuğberk (2020-11-13). "Neuropilin-1 facilitates SARS-CoV-2 cell entry and infectivity". Science. 370 (6518): 856–860. Bibcode:2020Sci...370..856C. doi:10.1126/science.abd2985. ISSN 0036-8075. PMC 7857391. PMID 33082293.
- ↑ 30.0 30.1 University of Helsinki (October 20, 2020). "Researchers discovered the second 'key' used by the SARS-CoV-2 virus to enter into human cells" (Press release). Eurekalert. Retrieved October 22, 2020.
- ↑ Doctor Duc C Vuong, general surgeon in Albuquerque, New Mexico (23 March 2020). "HOW COVID-19 KILLS--I'm a Surgeon--And Why We Can't Save You". YouTube. Retrieved 5 April 2020.
{{cite web}}: CS1 maint: multiple names: authors list (link) - ↑ Brigham and Women's Hospital (May 21, 2020). "Lungs of deceased COVID-19 patients show distinctive features" (Press release). Eurekalert.org.
{{cite press release}}: Text "https://www.eurekalert.org/pub_releases/2020-05/bawh-lod052120.php" ignored (help) - ↑ Maximilian Ackermann; Stijn E. Verleden; Mark Kuehnel; Axel Haverich; Tobias Welte; Florian Laenger; Arno Vanstapel; Christopher Werlein; Helge Stark; Alexandar Tzankov; William W. Li; Vincent W. Li; Steven J. Mentzer; Danny Jonigk (May 21, 2020). "Pulmonary Vascular Endothelialitis, Thrombosis, and Angiogenesis in Covid-19". New England Journal of Medicine. 383 (2): 120–128. doi:10.1056/NEJMoa2015432. PMC 7412750. PMID 32437596.
- ↑ "Symptoms of Coronavirus". Centers for Disease Control and Prevention. Retrieved May 4, 2020.
- ↑ "New COVID-19 guidance for gastroenterologists" (Press release). Eurekalert.org. May 4, 2020. Retrieved May 4, 2020.
- ↑ Amit Syal (October 1, 2020). "New research shows that pain relief from coronavirus may explain large asymptomatic crowd". Daily Wildcat. Archived from the original on October 5, 2020. Retrieved October 10, 2020.
- ↑ Wrapp, Daniel; Wang, Nianshuang; Corbett, Kizzmekia S.; Goldsmith, Jory A.; Hsieh, Ching-Lin; Abiona, Olubukola; Graham, Barney S.; McLellan, Jason S. (2020-03-13). "Cryo-EM structure of the 2019-nCoV spike in the prefusion conformation". Science (New York, N.Y.). 367 (6483): 1260–1263. Bibcode:2020Sci...367.1260W. doi:10.1126/science.abb2507. ISSN 0036-8075. PMC 7164637. PMID 32075877.
- ↑ "ArticleStructural and Functional Basis of SARS-CoV-2 Entryby Using Human ACE2" (PDF). Cell. 181 (4): 894–904.e9. March 14, 2020. doi:10.1016/j.cell.2020.03.045. PMC 7144619. PMID 32275855. Retrieved April 15, 2020.
{{cite journal}}: Cite uses deprecated parameter|authors=(help) - ↑ Renhong Yan; Yuanyuan Zhang; Yaning Li; Lu Xia; Yingying Guo; Qiang Zhou (March 27, 2020). "Structural basis for the recognition of SARS-CoV-2 by full-length human ACE2". Science. 367 (6485): 1444–1448. Bibcode:2020Sci...367.1444Y. doi:10.1126/science.abb2762. PMC 7164635. PMID 32132184.
- ↑ Jon Gambrell (March 22, 2020). "Iran leader refuses US help, citing virus conspiracy theory". Associated Press. Retrieved March 29, 2020.
- ↑ Katherine Schaeffer (April 8, 2020). "Nearly three-in-ten Americans believe COVID-19 was made in a lab". Pew Research. Retrieved April 17, 2020.
- ↑ Poppy Noor (April 13, 2020). "A third of Americans believe Covid-19 laboratory conspiracy theory – study". The Guardian. Retrieved April 17, 2020.
- ↑ Kristian G. Anderson; Andrew Rambaut; W. Ian Lipkin; Edward C. Holmes; Robert F. Garry (March 17, 2020). "The proximal origin of SARS-CoV-2". Nature Medicine. 26 (4): 450–452. doi:10.1038/s41591-020-0820-9. PMC 7095063. PMID 32284615.
- ↑ He, Feng; Deng, Yu; Li, Weina (July 2020). "Coronavirus disease 2019: What we know?". Journal of Medical Virology. 92 (7): 719–725. doi:10.1002/jmv.25766. ISSN 0146-6615. PMC 7228340. PMID 32170865.
- ↑ Zhang, Qi; Wang, Yakun; Qi, Changsong; Shen, Lin; Li, Jian (June 2020). "Clinical trial analysis of 2019-nCoV therapy registered in China". Journal of Medical Virology. 92 (6): 540–545. doi:10.1002/jmv.25733. ISSN 0146-6615. PMC 7228274. PMID 32108352.
- ↑ Eun Kim, Geza Erdos, Shaohua Huang, Thomas W. Kenniston, Stephen C. Balmert, Cara Donahue Carey, V. Stalin Raje, Michael W. Epperly, William B. Klimstrad,Bart L. Haagmans, Emrullah Korkmaz, Louis D. Falo Jr., and Andrea Gambotto (April 2, 2020). "Microneedle array delivered recombinant coronavirus vaccines:Immunogenicity and rapid translational development". eBioMedicine. 55: 102743. doi:10.1016/j.ebiom.2020.102743. PMC 7128973. PMID 32249203.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ↑ University of Pittsburgh (April 2, 2020). "COVID-19 vaccine candidate shows promise in first peer-reviewed research". Eurekalert. Retrieved April 3, 2020.
- ↑ University of British Columbia. "Trial drug can significantly block early stages of COVID-19 in engineered human tissues". Eurekalert. Retrieved April 5, 2020.
- ↑ Vanessa Monteil, Hyesoo Kwon, Patricia Prado, Astrid Hagelkrüys, Reiner A. Wimmer, Martin Stahl, Alexandra Leopoldi, Elena Garreta, Carmen Hurtado del Pozo, Felipe Prosper, J.P. Romero, Gerald Wirnsberger, Haibo Zhang, Arthur S. Slutsky, Ryan Conder, Nuria Montserrat, Ali Mirazimi, Josef M. Penninger (April 2, 2020). "Inhibition of SARS-CoV-2 infections in engineered human tissues using clinical-grade soluble human ACE2" (PDF). Cell. 181 (4): 905–913.e7. doi:10.1016/j.cell.2020.04.004. PMC 7181998. PMID 32333836. Retrieved April 5, 2020.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ↑ Bill Bostock (April 27, 2020). "6 monkeys given an experimental coronavirus vaccine from Oxford did not catch COVID-19 after heavy exposure, raising hopes for a human vaccine". Business Insider. Retrieved April 28, 2020.
- ↑ David D. Kirkpatrick (April 27, 2020). "In Race for a Coronavirus Vaccine, an Oxford Group Leaps Ahead". New York Times. Retrieved April 28, 2020.
- ↑ 52.0 52.1 Elizabeth Cohen (May 18, 2020). "Early results from Moderna coronavirus vaccine trial show participants developed antibodies against the virus". CNN. Retrieved May 19, 2020.
- ↑ Alexandra Sternlicht (May 19, 2020). "Scientists Raise Questions About Moderna Vaccine In Market-Shaking Report". Forbes. Retrieved May 19, 2020.
- ↑ Helen Branswell (May 19, 2020). "Vaccine experts say Moderna didn't produce data critical to assessing Covid-19 vaccine". STAT. Retrieved May 19, 2020.
- ↑ 55.0 55.1 Michael Crowley; Katie Thomas; Maggie Haberman (April 5, 2020). "Ignoring Expert Opinion, Trump Again Promotes Use of Hydroxychloroquine". New York Times. Retrieved April 6, 2020.
- ↑ 56.0 56.1 Peter Baker; Katie Rogers; David Enrich; Maggie Haberman (April 6, 2020). "Trump's Aggressive Advocacy of Malaria Drug for Treating Coronavirus Divides Medical Community". New York Times. Retrieved April 7, 2020.
- ↑ Richard Luscombe (April 6, 2020). "Fauci: no evidence anti-malaria drug Trump pushes works against virus". The Guardian. Retrieved April 6, 2020.
- ↑ BMJ (May 14, 2020). "Further evidence does not support hydroxychloroquine for patients with COVID-19" (Press release). Eurekalert. Retrieved May 18, 2020.
- ↑ 59.0 59.1 59.2 "Compassionate Use of Remdesivir for Patients with Severe Covid-19". New England Journal of Medicine. 382 (24): 2327–2336. April 10, 2020. doi:10.1056/NEJMoa2007016. PMC 7169476. PMID 32275812.
{{cite journal}}: Cite uses deprecated parameter|authors=(help) - ↑ 60.0 60.1 60.2 "Two-Thirds of Severe Covid-19 Cases Improved on Gilead Drug". Bloomberg News. Retrieved April 11, 2020.
{{cite news}}: Cite uses deprecated parameter|authors=(help) - ↑ Gina Kolata; Katherine J. Wu (October 15, 2020). "Remdesivir Fails to Prevent Covid-19 Deaths in Huge Trial". New York Times. Retrieved October 21, 2020.
Other websites[change | change source]
| Definitions from Wiktionary | |
| Media from Commons | |
| News stories from Wikinews | |
| Quotations from Wikiquote | |
| Source texts from Wikisource | |
| Textbooks from Wikibooks | |
| Travel guide from Wikivoyage | |
| Learning resources from Wikiversity | |
